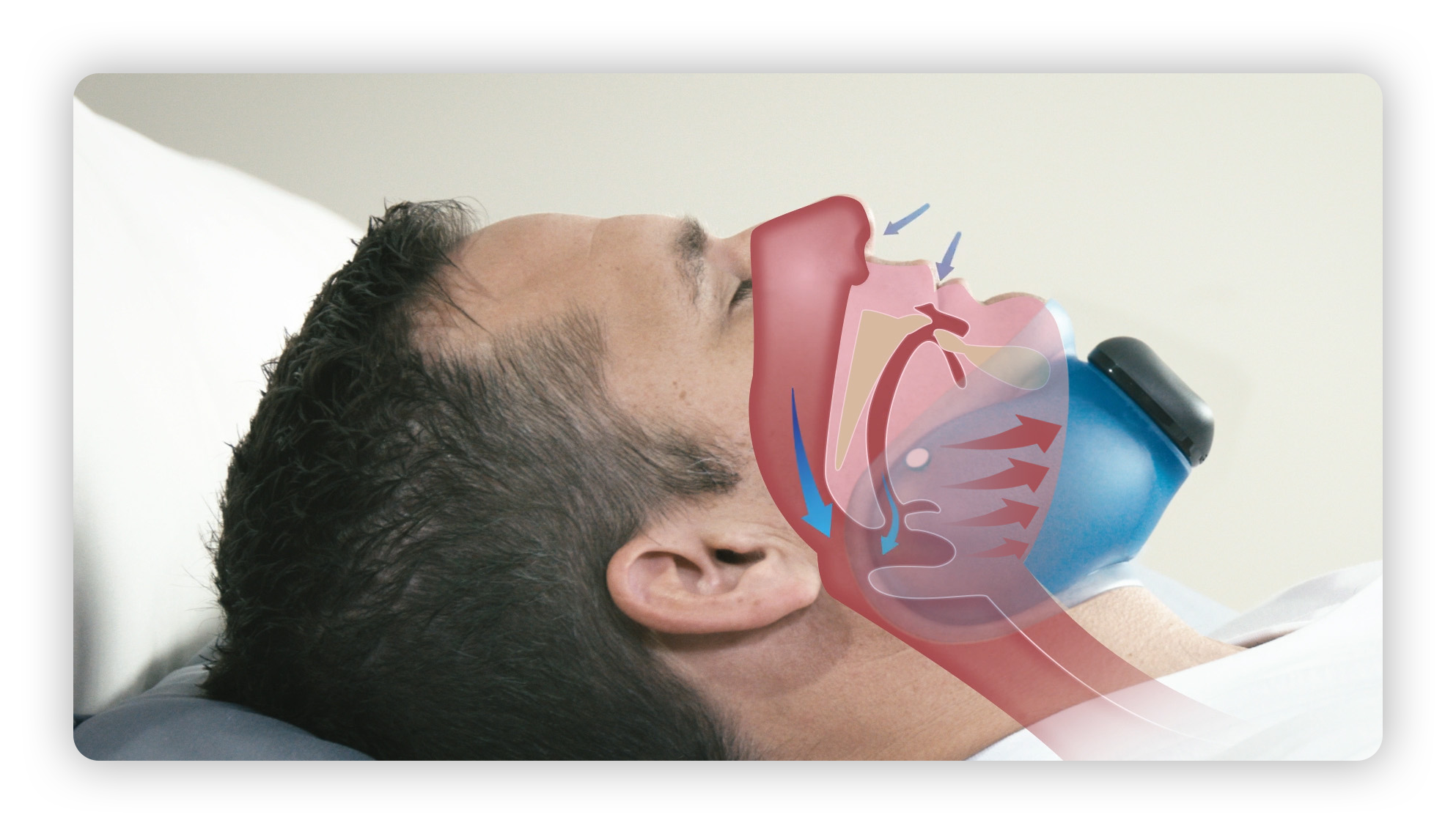
aerSleep™ is the first product for sleep apnea which is completely non-invasive and does not require connecting tubes or retaining straps. aerSleep™ really does allow you to sleep without limitations. (This product is not currently available in the U.S.)
How It Works
Benefits
- Quiet
- Small and Portable
- Simple: No masks, machine, hoses, cords, or humidifiers required
- Non-Invasive: Applied externally to the airway eliminating invasive treatments
- Easy to Use: Quickly set up in seconds for comfortable treatment
- Effective: Reduces the number of apnea and oxygen desaturation events1
aerSleep consists of a comfortable, flexible silicone rubber collar and a small, lightweight, quiet air pump that automatically regulates and maintains therapy. Using it is easy. Simply turn it on, place it under your chin and go to sleep.
aerSleep uses aer+TM technology (formerly known as cNEP), which is our patented technology that uses the application of negative external air pressure on the outside of the neck to gently open the airway.
aerSleep is a simple to use, non-invasive external airway device that effectively and safely reduces the number of apnea and oxygen desaturation events.
A clinical study was conducted at the Toronto Sleep Institute in Toronto, Ontario, Canada, utilizing aerSleep to treat patients with mild, moderate or severe OSA, and measuring efficacy through two in-lab polysomnographs (PSG).
Conclusion: aerSleep was found to be both safe and effective in delivering treatment to subjects with mild, moderate or severe OSA.
Study Results
- The frequency of apnea and low oxygen levels were significantly reduced compared to baseline values
- Frequency of apneas and hypopneas reduced by 74% in those responsive to aerSleep
- Episodes of oxygen desaturation reduced by 81%
- Home use of aerSleep was well tolerated
Patient Responses
- 70% of patients stated that aerSleep would be acceptable for long-term treatment of their sleep apnea.
- 88% of patients stated their experience was better compared to previous treatment methods.
- 82% of patients stated their sleep was better using aerSleep compared to their usual night’s sleep.
Additional Clinical Studies

“Evaluation of Continuous Negative External Pressure (cNEP) for the Treatment of Obstructive Sleep Apnea: A Pilot Study.” August 2017. J Kram et al.
- Subjects with documented OSA were recruited from the patient population at one sleep clinic. The intervention was application and titration of cNEP during overnight PSG. cNEP appeared to be safe and effective during short-term use in subjects with OSA.
Read more at ncbi.nlm.nih.gov/pubmed/28633720

“Submental negative pressure application decreases collapsibility of the passive pharyngeal airway in nonobese women.” April 2015. Kato S et al.
- Negative external pressure (NEP) application to the submandible region improves pharyngeal airway collapsibility. Conclusively, application of submental negative pressure was found to decrease collapsibility of the passive pharyngeal airway in nonobese Japanese women.
Read more at ncbi.nlm.nih.gov/pubmed/25614595
* aerSleep™ is not currently available in the U.S.
1 SOM-015 Clinical Study Report